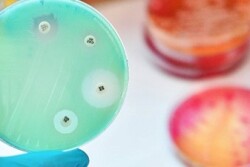
Antibiotic Resistance
With food recalls skyrocketing, one might find it hard to discern whether they’re reading a current USDA Food Safety and Inspection Service report or a chapter straight out of Upton Sinclair’s 1906 novel “The Jungle.”
USDA’s APHIS is awarding more than $3.2 million to create antimicrobial resistance dashboards to improve access to information on antimicrobial resistance in domesticated animals.
While another month has come and gone for the 2023 production year, if you haven’t already heard, June will hold some significant changes for the livestock industry in the area of antibiotics.
On June 11, FDA’s Guidance for Industry #263 brings 91 over-the-counter antimicrobial products from OTC to prescription oversight. Three experts weigh in on why you need to prepare for this change now.
The International Consortium for Antimicrobial Stewardship in Agriculture (ICASA) has awarded nine grants to develop management strategies that improve judicious antibiotic use in beef cattle and swine.
While several brand-name and generic products are available at a variety of price points, what questions do you ask yourself when there’s a sick animal needing care?
USDA’s National Institute of Food and Agriculture announced an investment of more than $5 million to mitigate antimicrobial resistance across the food chain.
Probiotics may not be as beneficial for animal and human health as people think, according to recently published research at Kansas State University. Here’s why.
Antimicrobial resistance is a “One Health” challenge that requires effort across both human and animal health sectors. Animal health companies have invested billions as part of a strategy to reduce antibiotic use.
A new report,“Beyond Antibiotics: The Future of Animal Health Alternatives,” identifies alternatives that could help support the animal health industry and producers, while reducing antibiotic use.
This week, the National Antimicrobial Resistance Monitoring System (NARMS) released its 2016-2017 NARMS Integrated Summary.
A pending trade agreement between the US and the EU would give the US 50% of the EU’s imported beef.
According to an analysis published in the journal Science, antibiotic resistance among bacteria affecting food animals has nearly tripled over the past 20 years.
The FDA has awarded two $250,000 grants to fund research projects in fiscal year 2019 to help target and define durations of use for certain medically important antimicrobial drugs administered in animal feed.
Veterinarians and livestock producers have adapted to significant changes in antimicrobial regulations, and change will continue as the FDA works through its five-year plan for antimicrobial stewardship.
In a release earlier this month, the U.S. Food and Drug Administration announced approval of Monovet 90, the first generic monensin product for use in cattle and goats.
Composting can reduce the concentration of antibiotics in cattle manure, but the degree of that reduction can vary depending on how it is tested.
The complex challenges of addressing antimicrobial resistance (AMR) make it a prototypical “One-Health” issue, according to five new papers published in the Annals of the New York Academy of Sciences.
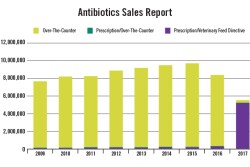
Report shows substantial decline in sales of medically important antibiotics for food animal use during 2017.
Noting the importance of keeping the use of antimicrobials as low as possible without compromising health, three international veterinary organizations have issued a statement regarding judicious use in food animals.
Study indicates same levels of resistant pathogens in ground beef raised with and without antibiotics.
Veterinarians will, over the next five years, become more involved in overseeing the use of all forms of medically important antimicrobials used in livestock.
When new veterinary feed directive (VFD) rules launched last year, FDA indicated they would initially focus on education, rather than penalties, to facilitate adoption and compliance.
A Texas Tech researcher has received funding from both USDA and NCBA for a project to evaluate the distribution of antimicrobial resistance (AMR) genes and the occurrence of horizontal gene transfer in high-risk cattle.
McDonald’s has announced plans to partner with its global beef suppliers to reduce the use of medically important antibiotics by the end of 2020.