Merck Animal Health Expands Voluntary Recall of Banamine
The company announced on Friday, Sept. 29, it is voluntarily recalling four additional batches of Banamine/Banamine-S (flunixin meglumine injection) 50 mg/mL in the United States, used for injection in cattle, swine and horses due to the presence of particulate matter.
Merck chose to make the recall following its observation of particulate matter in specific product batches during routine quality testing, according to a press release the company issued.
This latest step follows a separate recall Merck made on Sept. 1 for three batches of Banamine/Banamine-S Injection.
What Are Particulates?
The FDA defines particulates as “mobile, undissolved particles (metal, glass, dust, fiber, rubber, polymer, mold, degradant precipitate) other than gas bubbles that are unintentionally present in an injectable product.”
The administration of an injectable product that contains particulate matter may result in local irritation, swelling or infection in response to the foreign material. After intravenous administration in large animals, such as cattle or horses, particulate matter could travel to the lungs which could result in local tissue damage.
The branded flunixin meglumine injection from Merck is approved in the U.S. only for intravenous use in beef and dairy cattle, and for intramuscular use in swine and for intravenous and intramuscular use in horses.
Four Specific Batches Of Product
The latest recall is for the following four batches of product:
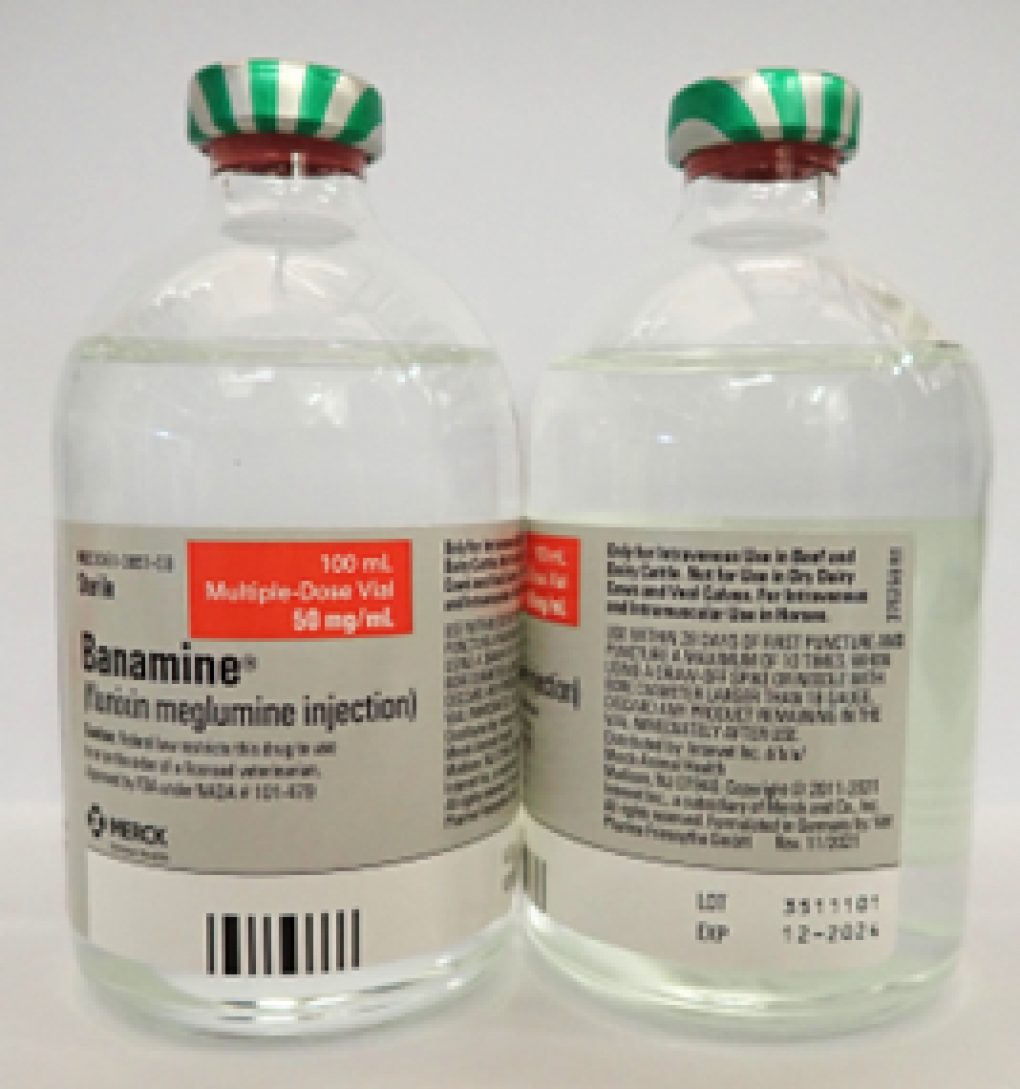
BANAMINE 100mL, UIN 065474, NDC 00061-0851-03, Batch 3511101, expires December 2024 Distribution dates: May 16, 2023, to Aug. 8, 2023
BANAMINE 100mL, UIN 065474, NDC 00061-0851-03, Batch 3511104, expires December 2024
Distribution dates: Aug. 4-17, 2023
BANAMINE 250mL, UIN 065476, NDC 00061-0851-04, Batch 3522101, expires December 2024
Distribution dates: July 14-Aug. 17, 2023
BANAMINE-S 100mL, UIN 065477, NDC 0061-1838-30, Batch 3511103, expires December 2024
Distribution dates: May 3-Aug.16, 2023
The lot number (LOT) and expiry date (EXP) are located in the bottom right portion of the bottle labels. For reference, see the photo of a bottle of Banamine below.
Next Steps To Consider
Merck says veterinarians, along with other customers, who have received Banamine and Banamine-S from the batches being recalled should stop using the products and refer to their recall letter for product return instructions.
Merck Animal Health is working with distributors to ensure that unused product is no longer in distribution or with customers. The company is notifying its distributors and customers directly and arranging for the return of all recalled product.
Consumers with technical questions regarding this recall should call 1-800-221-3573 (8 a.m. – 5 p.m. weekdays Central Time). Customers who may need to arrange return of product should contact their point of purchase.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA at 1-888-FDA-VETS or online.
This recall is being made with the knowledge of the Food and Drug Administration